Product Introduction
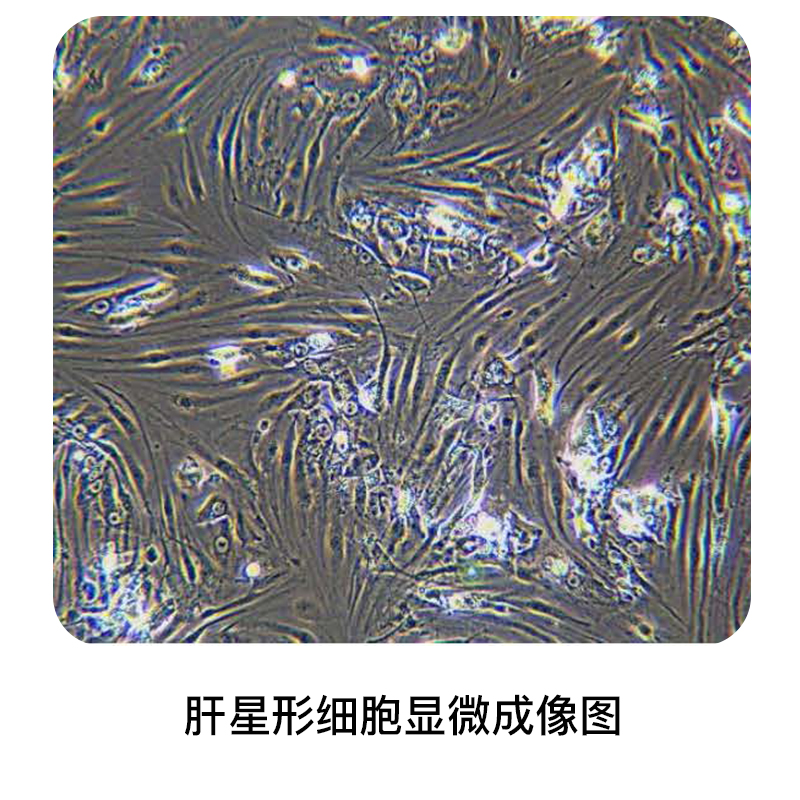
Hepatic Stellate Cell
(Plateable,Cat# LV-HSC001/2/3/4)
(For research use only)
In order to ensure the safety and biosafety of the experimenters, please wear necessary protective equipment such as protective masks, latex gloves and protective eye shields (during revival) when contacting this product and its waste. Please operate in strict accordance with this manual, and the waste generated after the experiment must be harmless treated in accordance with the relevant laws and regulations to ensure biological safety.
Ⅰ. Introduction
Hepatic stellate cells (HSC) account for about 30% of non-parenchymal cells. HSC is present in the Disse cavity, spindle or polygonal, with multiple vitamin A-rich lipid droplets in the cytoplasm. Its elongated protrusion extends outward around the sinusoidal endothelial cells, and it is the primary storage of retinoid derivatives in the body. In normal liver, HSC are quiescent, do not express α smooth muscle actin (α-SMA), have low proliferative activity and low capacity to synthesize collagen, and their main function is to store retinoid aldehydes.
Ⅱ. Reagents and Materials
- Hepatic stellate cell (Cat# LV-HSC001/2/3/4)
- Recovery medium(Cat#LV-Rec001)
- Stellate cell culture medium (Cat# LV-HSCM001)
- Collagen-coated plate (Cat# LV-Coated)
- Crushed ice and ice boxes
- Sterile centrifuge tube of 15 ml
- Disposable pipette
- Wide-mouth pipette tip (Cut off the tip of normal pipette and sterilize it.)
- Pipette
-Thermostatic water bath (Preheat at 38℃)
-Low-temperature horizontal centrifuge (with a horizontal rotor, it is possible to centrifuge a 15ml centrifuge tube)
- Biosafety cabinet
- 37 °C/5% CO2 incubator
- 75% alcohol
Ⅲ. Recovery and Plating of Cells
1. Insert the 15 ml centrifuge tube into an ice box containing enough crushed ice and sterilize it in UV for 15 min. Pre-cool the centrifuge.
2. The recovery medium should be placed in crushed ice for sufficiently precooling, and the plating medium should be placed in a 38 °C thermostatic water bath to be fully preheated.
3. Quickly transfer the frozen cells from the refrigerated position to a thermostatic water bath of 38℃. Then,immerse them in as much water as possible at 38°C and shake clockwise. Please make sure that the cap of the cryogenic tube is kept above the water.
4. Thaw the freezing tube for about 90-120s, until only a small amount of crushed ice floats in it.
5. Sterilize the freezing tube with 75% alcohol and transfer it to a biosafety cabinet.
6. Resuspend the cells with a wide-mouth pipet tip (gently blow 2 times) and transfer to a pre-cooled centrifuge tube of 15 ml.(Note: if there are more cells left on the freezing tube and the pipette tip, aspirate 1 ml of recovery medium to rinse them.
7. Add pre-cooled recovery medium to the cell suspension dropwise and add 10 ml of recovery medium to every 1 ml of cell suspension (Note: the first 3 ml should be added dropwise and shaken slightly, and the next 7 ml can be accelerated). Finally, slightly upside down 1 time to mix well.
8. Centrifuge 600×g at 4 °C for 5 min, remove the supernatant and resuspend with plating medium.
9. The pelleted cells should be resuspended with hepatic stellate cell culture medium and set the volume to 4 ml. The viability and total amount of hepatic stellate cell can be determined by Trypan Blue Exclusion Method.
10. Seed the living cells at the density of 3×104cells/cm2, shake well and culture in a 37 °C/5% CO2 incubator. Change the solution after 24h.IV. Culture and Passage of Hepatic Stellate Cell
1. Cells can be passaged at 80% confluence.
2. Preheat the culture medium, PBS and pancreatic enzyme into a 37℃ water bath in advance, wipe it with 75% alcohol, and then put it into the ultra-clean table.
3. Aspirate the old culture medium, add a small amount of PBS. After discarding PBS, add an amount of trypsin, so that the added trypsin could cover the cells. Incubate at 37℃, and observe at about 2~3min under the microscope.
4. When the space between the plateable cells becomes larger and the cells become round but not yet floating, discard the pancreas. Add fresh medium, shake the cell bottle, and stop the pancreatic enzyme. Use a straw carefully to blow the plateable cells to make cell suspension (control the intensity of blowing, to avoid a large number of bubbles).
5. Centrifuged at 600×g for 5min at room temperature, remove the supernatant and resuspend it with the culture medium.
6. Seed the cell suspension at the density of 3×104cells/cm2 onto a new TC-treated culture plate and incubate it in a 37℃/5% CO2 incubator. Observe the growth of plateable cells next day.
Ⅴ. Customer Service
If you find any quality problems with the product, please collect the original data and contact the company's salesmen or technical support at the first time. The company ensure after-sales service. Every laboratory has different conditions, different operating habits, different proficiency, and objective factors in experimental failure. If the operation is not strictly in accordance with the instructions or exceeds the time limit of after-sales, the company does not do after-sales. Please understand and support us.
Validity period and raw data provided:
Resuscitation problems: Within 24 hours of resuscitation, trypan blue staining or PI staining should be provided.
Pollution problems: Within 96 hours of resuscitation, microscope photographs of differences should be provided.
Purity issues: Within one month, immunofluorescence or flow cytometry results should be provided.
VI. Contact Number
Tel: 0755-28284050
Technical Support: 19902901483 (Dr. Zhou)