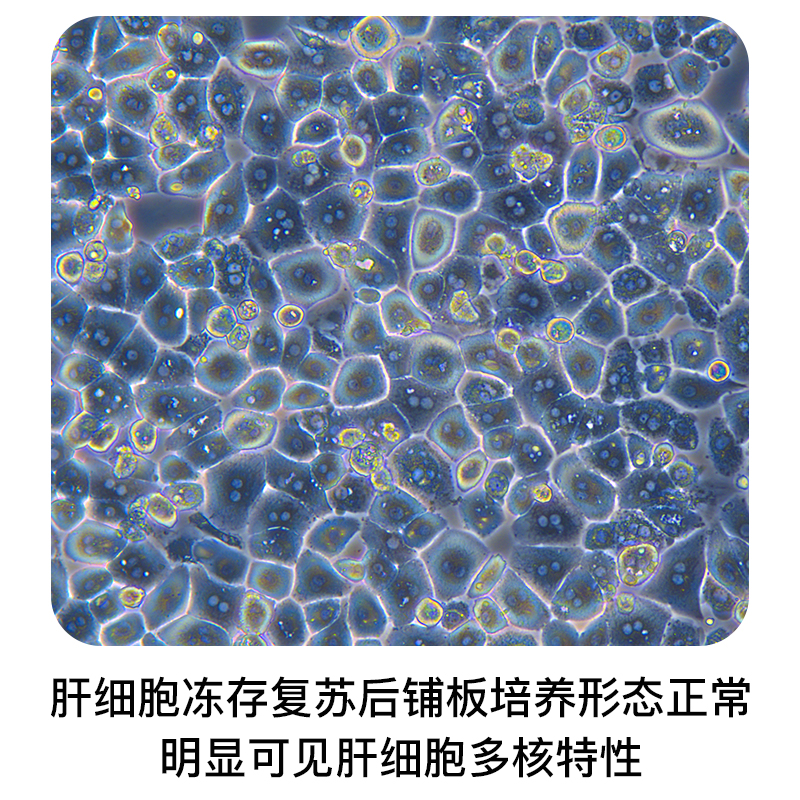
Product Introduction
|
Product |
Primary Monkey Hepatocytes |
|
Catalog |
LV-PMonH001 |
|
Spec. |
4-8million |
|
Strains |
Cynobial monkeys/Macaques |
|
Number of donors |
N=1,4 |
|
Free shipping or not |
NO |
|
Delivery |
1week |
|
Carriage |
Liquid nitrogen |
|
Storage |
Liquid nitrogen |
|
Life cycle |
10years |
|
Warranty |
7years |
|
|
Primary Monkey Hepatocytes
(Cat# LV-PMonH001/2)
(For research use only)
In order to ensure the safety and biosafety of the experimenters, please wear necessary protective equipment such as protective masks, latex gloves and protective eye shields (during revival) when contacting this product and its waste. Please operate in strict accordance with this manual, and the waste generated after the experiment must be harmless treated in accordance with the relevant laws and regulations to ensure biological safety.
I. Introduction
Liver-Biotech is a national high-tech enterprise focusing on the isolation, cryopreservation and regeneration of primary hepatocytes. Primary monkey hepatocytes are isolated from clean grade or SPF monkeys with high cell purity (>95%). The main species are cynomolgus monkeys and rhesus monkeys. After a variety of tests, the recovery cells are highly viable and polarized (differentiated), and could be widely used in basic life science research and drug discovery.
II. Reagents and Materials
- Primary monkey hepatocytes (Cat# LV-PMonH001/2)
- Recovery medium (Cat#LV-Rec001/3)
- Purification medium (provide if needed)
- Plating medium
(Plateable cells are needed. Cat#LV-WEP007)
- Maintenance medium
(Plateable cells are needed. Cat#LV-WEM007)
- Collagen-coated plate
(Plateable cells are needed. Cat #LV-Coated)
- Sterile centrifuge tube of 15ml
- Disposable pipette
- Wide-mouth pipette tip (Cut off the tip of normal pipette and sterilize it.)
- Pipette
- Thermostatic water bath (Preheat at 37℃. Please calibrate the temperature with a thermometer)
- Low-temperature horizontal centrifuge (with a horizontal rotor, it is possible to centrifuge a 15ml centrifuge tube)
- Biosafety cabinet
- 37 °C/5% CO2 incubator
- 75% alcohol
III. Resurgence of Suspension Cells
1. Add 10ml recovery medium (Cat#LV-Rec003) to a 15ml centrifuge tube in a biosafety cabinet, pre-warm a thermostatic water bath at 37°C for 20 min, and then transfer the medium to a biosafety cabinet, and pre-warm the plating medium at 37°C.
2. Rapidly transfer the frozen hepatocytes by a liquid-nitrogen thermos from the refrigerated location to a 37°C thermostatic water bath and immerse as much as possible in 37°C water with clockwise horizontal rolling, but must ensure that the cap of cryopreservation tube stays above the water.
3. Thaw the cryopreservation tube for about 90 ~120s until only a small piece of crushed ice floats in it.
4. Sterilize the cryopreservation tube with 75% alcohol and transfer it to a biosafety cabinet.
5. Aspirate the cells with a wide-mouth pipette tip and add them dropwise to a 15ml centrifuge tube containing the preheated recovery medium. (Note: there are some cells left on the cryopreservation tube and the pipette tip, aspirate 1ml recovery medium to rinse. Then, transfer the 1ml recovery medium into the 15ml centrifuge tube). Slightly turn the 15ml centrifuge tube upside down 2-3 times to mix cells well.
6. Centrifuge 50×g of the 15 ml centrifuge tube at low speed (Higher centrifugation speed may has an adverse effect on cell viability), at room temperature for 5min. After the supernatant being removed, resuspend cells with a metabolism medium (provided by the customer) for drug metabolism, and the viability and number of viable hepatocytes were determined by Trypan Blue Exclusion Method (manual counting on a hemocytometer, not recommend the cell counter machine). Add the drug according to the experimental requirements and measure the metabolites.
IV. Recovery of Plateable Cells
1. In the biosafety cabinet, add 10ml of recovery medium (Cat#LV-Rec001) to a 15ml centrifuge tube and preheat in thermostatic water bath of 37 °C for 20 min. Then transfer it to the biosafety cabinet. Purification medium should be placed at room temperature (if necessary); preheat plating medium at 37 °C.
2. Rapidly transfer the frozen hepatocytes by a liquid-nitrogen thermos from the refrigerated location to a 37°C thermostatic water bath and immerse as much as possible in 37°C water with clockwise horizontal rolling, but must ensure that the cap of cryopreservation tube stays above the water.
3. Thaw the cryopreservation tube for about 90-120s, until only a small amount of crushed ice floats in it.
4. Sterilize the cryopreservation tube with 75% alcohol and transfer it to a biosafety cabinet.
5. Aspirate the cells with a wide-mouth pipette tip and add them dropwise to a centrifuge tube containing the preheated recovery medium. (Note: there are more cells left on the cryopreservation tube and the pipette tip, aspirate 1ml recovery medium to rinse. Then, mix the 1ml recovery medium into the centrifuge tube medium). Slightly turn the tube upside down 2-3 times to mix them well.
6. Directly centrifuged 50×g of 15ml centrifuge tube at a low speed (Higher centrifugation speed may has an adverse effect on cell viability) and at room temperature for 5 min. After the supernatant being removed, resuspend cells with 4ml of plating medium. The viability of hepatocytes can be measured with Trypan Blue Exclusion Method (manual counting on a hemocytometer, not recommend the cell counter machine). If the viability rate is more than 60%, you can directly plate onto the collagen-coated plate, otherwise purify the hepatocytes before plating.
7. Cell purification (optional step): Centrifuge hepatocytes suspension at 50×g, at room temperature, for 5 min. Remove the supernatant and resuspend the cells with 2ml of purification medium. Then, carefully adds 1ml plating medium above purification medium along the centrifuge tube wall to the centrifuge tube (Do not stir the lower layer, obvious stratification happens after adding the plating medium). Centrifuge at 800×g at 4°C for 20 min (set maximum acceleration to 9, set minimum deceleration to 1). After centrifugation, aspirate the turbid layer (living cell layer) between the purification medium and the plating medium, and transfer the turbid layer to a new 15ml centrifuge tube. Mix twice volumes of plating medium and the turbid layer in the new 15ml centrifuge tube. After that, centrifuge 50×g of the 15ml centrifuge tube for 5 min at room temperature, remove the supernatant and resuspend the cells with 4ml plating medium.
8. Measure the viability and total number of living hepatocytes by Trypan Blue Exclusion Method (manual counting on a hemocytometer, not recommend the cell counter machine). It is recommended to repeat 3 times to get an average.
9. Seed the living cells at the density of 1.2-1.5×105cells/cm2 onto collagen-coated plate. Shake the plate well and incubate it in a 37℃/5% CO2 incubator. Recommended step: 4h post seeding, gently change the culture medium (at this time, the cells may be in a semi-adherent state) by adding freshly preheated plating medium to improve the cells state.
V. Maintenance of Hepatocyte
1. After culturing the hepatocytes for 8 hours or overnight (counting from the start of seeding), the cells have been well seeded. Remove the plating medium (containing part of the dead cells), add the maintenance medium, and change the culture medium every 2 days.
2. Primary monkey hepatocytes have limited proliferation capacity and obvious characteristics of contact inhibition; the differentiation characteristics of hepatocytes and the maintenance time all depend on high-density cell culture.
The maintenance time of primary monkey hepatocytes in vitro is relatively short. The duration of the experiment is recommended to be completed within 7 days and up to 9 days after plating. It is not recommended to subculture the primary monkey hepatocytes
VI. Customer Service
If you find any quality problems with the product, please collect the original data and contact the company's salesmen or technical support at the first time. The company ensure after-sales service. Every laboratory has different conditions, different operating habits, different proficiency, and objective factors in experimental failure. If the operation is not strictly in accordance with the instructions or exceeds the time limit of after-sales, the company does not do after-sales. Please understand and support us.
Validity period and raw data provided:
Resuscitation problems: Within 24 hours of resuscitation, trypan blue staining or PI staining should be provided.
Pollution problems: Within 96 hours of resuscitation, microscope photographs of differences should be provided.
Purity issues: Within one month, immunofluorescence or flow cytometry results should be provided
VII. Contact Number
Tel:0755-28284050
Technical Support: 19902901483 (Dr. Zhou)